| Entry |
|
|---|
| Name |
Cetirizine hydrochloride (JP18/USP);
Zyrtec (TN) |
|---|
| Product |
|
|---|
| Generic |
CETIRIZINE HYDROCHLORIDE (A-S Medication Solutions), CETIRIZINE HYDROCHLORIDE (A-S Medication Solutions), CETIRIZINE HYDROCHLORIDE (Amneal Pharmaceuticals LLC), CETIRIZINE HYDROCHLORIDE (Aventura Pharmaceuticals), CETIRIZINE HYDROCHLORIDE (BAJAJ MEDICAL), CETIRIZINE HYDROCHLORIDE (Breckenridge Pharmaceutical), CETIRIZINE HYDROCHLORIDE (Bryant Ranch Prepack), CETIRIZINE HYDROCHLORIDE (DIRECT RX), CETIRIZINE HYDROCHLORIDE (PAI Holdings), CETIRIZINE HYDROCHLORIDE (Padagis Israel Pharmaceuticals), CETIRIZINE HYDROCHLORIDE (Preferred Pharmaceuticals), CETIRIZINE HYDROCHLORIDE (Preferred Pharmaceuticals), CETIRIZINE HYDROCHLORIDE (Proficient Rx LP), CETIRIZINE HYDROCHLORIDE (Proficient Rx LP), CETIRIZINE HYDROCHLORIDE (Taro Pharmaceuticals U.S.A.), CETIRIZINE HYDROCHLORIDE ORAL SOLUTION (Chartwell RX) |
|---|
| Formula |
C21H25ClN2O3. 2HCl
|
|---|
| Exact mass |
460.1087
|
|---|
| Mol weight |
461.8097
|
|---|
| Structure |
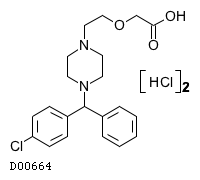
|
|---|
| Simcomp |
|
|---|
| Class |
Anti-allergic agent
DG01557 Histamine receptor antagonist
DG01482 Histamine receptor H1 antagonist
Dermatological agent
DG03243 Atopic dermatitis agent
Transporter substrate
DG01665 ABCB1 substrate
DG02854 SLC22A2 substrate
Transporter inhibitor
DG02863 SLC22A2 inhibitor
|
|---|
| Remark |
| Therapeutic category: | 4490 |
| Product (DG01107): | D00664<JP/US> |
|
|---|
| Efficacy |
Antiallergic, H1 receptor antagonist |
|---|
| Target |
|
|---|
| Pathway |
| hsa04080 | Neuroactive ligand-receptor interaction |
| hsa04750 | Inflammatory mediator regulation of TRP channels |
|
|---|
| Metabolism |
Transporter: ABCB1 [HSA: 5243], SLC22A2 [HSA: 6582]
|
|---|
| Interaction |
Transporter inhibition: SLC22A2 [HSA: 6582]
|
|---|
| Structure map |
| map07212 | Histamine H1 receptor antagonists |
|
|---|
| Brite |
Anatomical Therapeutic Chemical (ATC) classification [BR:br08303]
R RESPIRATORY SYSTEM
R06 ANTIHISTAMINES FOR SYSTEMIC USE
R06A ANTIHISTAMINES FOR SYSTEMIC USE
R06AE Piperazine derivatives
R06AE07 Cetirizine
D00664 Cetirizine hydrochloride (JP18/USP) <JP/US>
S SENSORY ORGANS
S01 OPHTHALMOLOGICALS
S01G DECONGESTANTS AND ANTIALLERGICS
S01GX Other antiallergics
S01GX12 Cetirizine
D00664 Cetirizine hydrochloride (JP18/USP) <JP/US>
USP drug classification [BR:br08302]
Ophthalmic Agents
Ophthalmic Anti-allergy Agents
Antihistamine
Cetirizine
D00664 Cetirizine hydrochloride (JP18/USP)
Respiratory Tract/Pulmonary Agents
Antihistamines
Histamine (H1) Receptor Antagonists, Mildly/Non-sedating
Cetirizine
D00664 Cetirizine hydrochloride (JP18/USP)
Therapeutic category of drugs in Japan [BR:br08301]
4 Agents affecting cellular function
44 Allergic agents
449 Miscellaneous
4490 Miscellaneous
D00664 Cetirizine hydrochloride (JP18/USP)
Classification of Japanese OTC drugs [BR:br08313]
Agents for otolaryngologic use
69 Oral medicine for nasal inflammation
D00664 Cetirizine hydrochloride (JP18/USP)
Risk category of Japanese OTC drugs [BR:br08312]
Second-class OTC drugs
Inorganic and organic chemicals
Cetirizine
D00664 Cetirizine hydrochloride (JP18/USP)
Drug groups [BR:br08330]
Anti-allergic agent
DG01557 Histamine receptor antagonist
DG01482 Histamine receptor H1 antagonist
DG01107 Cetirizine
D00664 Cetirizine hydrochloride
Dermatological agent
DG03243 Atopic dermatitis agent
D00664 Cetirizine hydrochloride
Transporter substrate
DG01665 ABCB1 substrate
DG01107 Cetirizine
D00664 Cetirizine hydrochloride
DG02854 SLC22A2 substrate
DG01107 Cetirizine
D00664 Cetirizine hydrochloride
Transporter inhibitor
DG02863 SLC22A2 inhibitor
DG01107 Cetirizine
D00664 Cetirizine hydrochloride
Drug classes [BR:br08332]
Anti-allergic agent
DG01482 Histamine receptor H1 antagonist
D00664 Cetirizine hydrochloride
Dermatological agent
DG03243 Atopic dermatitis agent
D00664 Cetirizine hydrochloride
Target-based classification of drugs [BR:br08310]
G Protein-coupled receptors
Rhodopsin family
Histamine
HRH1
D00664 Cetirizine hydrochloride (JP18/USP) <JP/US>
Drugs listed in the Japanese Pharmacopoeia [BR:br08311]
Chemicals
D00664 Cetirizine hydrochloride
D00664 Cetirizine hydrochloride tablets
Drug metabolizing enzymes and transporters [br08309.html]
Drug transporters
D00664
Rx-to-OTC switch list in the USA [br08315.html]
D00664
Rx-to-OTC switch list in Japan [br08314.html]
D00664
Drug groups [BR:br08330]
Anti-allergic agent
DG01557 Histamine receptor antagonist
DG01482 Histamine receptor H1 antagonist
DG01107 Cetirizine
Transporter substrate
DG01665 ABCB1 substrate
DG01107 Cetirizine
DG02854 SLC22A2 substrate
DG01107 Cetirizine
Transporter inhibitor
DG02863 SLC22A2 inhibitor
DG01107 Cetirizine
|
|---|
| Other DBs |
|
|---|
| LinkDB |
|
|---|
| KCF data |
ATOM 29
1 C8x C 22.3396 -18.4537
2 C8y C 22.3396 -19.8566
3 C8x C 23.5544 -20.5580
4 C8x C 24.7694 -19.8566
5 C8y C 24.7694 -18.4537
6 C8x C 23.5544 -17.7523
7 C8x C 27.1992 -19.8566
8 C8y C 27.1992 -18.4537
9 C1c C 25.9843 -17.7523
10 C8x C 28.4141 -20.5580
11 C8x C 29.6290 -19.8566
12 C8x C 29.6290 -18.4537
13 C8x C 28.4141 -17.7523
14 N1y N 25.9843 -16.3494
15 C1x C 27.2013 -15.6468
16 C1x C 27.2013 -14.2439
17 N1y N 25.9863 -13.5425
18 C1x C 24.7694 -14.2451
19 C1x C 24.7694 -15.6480
20 X Cl 21.1247 -20.5580
21 C1b C 25.9863 -12.1408
22 C1b C 27.2018 -11.4391
23 O2a O 28.3999 -12.1310
24 C1b C 29.5895 -11.4443
25 C6a C 30.7833 -12.1337
26 O6a O 31.9752 -11.4456
27 O6a O 30.7833 -13.5434
28 X Cl 31.6686 -16.0688
29 X Cl 31.6686 -16.0688
BOND 29
1 1 2 2
2 2 3 1
3 3 4 2
4 4 5 1
5 5 6 2
6 1 6 1
7 7 8 1
8 8 9 1
9 5 9 1
10 7 10 2
11 10 11 1
12 11 12 2
13 12 13 1
14 8 13 2
15 9 14 1
16 14 15 1
17 15 16 1
18 16 17 1
19 17 18 1
20 18 19 1
21 14 19 1
22 2 20 1
23 17 21 1
24 21 22 1
25 22 23 1
26 23 24 1
27 24 25 1
28 25 26 1
29 25 27 2
BRACKET 1 30.2400 -16.8000 30.2400 -15.2600
1 32.3400 -15.2600 32.3400 -16.8000
1 2
ORIGINAL 1 28
REPEAT 1 29
|
|---|