 | | DRUG: パントプラゾールナトリウム水和物 | |
| エントリ |
|
|---|
| 一般名 |
パントプラゾールナトリウム水和物 (JAN);
Pantoprazole sodium (USP);
Pantoprazole sodium hydrate (JAN) |
|---|
| 米国の商品 |
|
|---|
| 後発品 |
PANTOPRAZOLE (Advanced Rx of Tennessee), PANTOPRAZOLE (Dr.Reddy's Laboratories Limited), PANTOPRAZOLE (NorthStar RxLLC), PANTOPRAZOLE (NuCare Pharmaceuticals), PANTOPRAZOLE (NuCare Pharmaceuticals), PANTOPRAZOLE (NuCare Pharmaceuticals), PANTOPRAZOLE (PHOENIX RX LLC), PANTOPRAZOLE (Preferred Pharmaceuticals), PANTOPRAZOLE (ST. MARY'S MEDICAL PARK PHARMACY), PANTOPRAZOLE SODIUM (A-S Medication Solutions), PANTOPRAZOLE SODIUM (A-S Medication Solutions), PANTOPRAZOLE SODIUM (A-S Medication Solutions), PANTOPRAZOLE SODIUM (A-S Medication Solutions), PANTOPRAZOLE SODIUM (A-S Medication Solutions), PANTOPRAZOLE SODIUM (A-S Medication Solutions), PANTOPRAZOLE SODIUM (Advanced Rx Pharmacy of Tennessee), PANTOPRAZOLE SODIUM (Advanced Rx Pharmacy of Tennessee), PANTOPRAZOLE SODIUM (Advanced Rx Pharmacy of Tennessee), PANTOPRAZOLE SODIUM (Advanced Rx Pharmacy of Tennessee), PANTOPRAZOLE SODIUM (Advanced Rx of Tennessee), PANTOPRAZOLE SODIUM (Advanced Rx of Tennessee), PANTOPRAZOLE SODIUM (Ajanta Pharma USA), PANTOPRAZOLE SODIUM (Alembic Pharmaceuticals Limited), PANTOPRAZOLE SODIUM (Alembic Pharmaceuticals), PANTOPRAZOLE SODIUM (American Health Packaging), PANTOPRAZOLE SODIUM (American Health Packaging), PANTOPRAZOLE SODIUM (Amneal Pharmaceuticals LLC), PANTOPRAZOLE SODIUM (Aphena Pharma Solutions - Tennessee), PANTOPRAZOLE SODIUM (Aphena Pharma Solutions - Tennessee), PANTOPRAZOLE SODIUM (Aphena Pharma Solutions - Tennessee), PANTOPRAZOLE SODIUM (Asclemed USA), PANTOPRAZOLE SODIUM (Asclemed USA), PANTOPRAZOLE SODIUM (Asclemed USA), PANTOPRAZOLE SODIUM (Aurobindo Pharma Limited), PANTOPRAZOLE SODIUM (Aurobindo Pharma Limited), PANTOPRAZOLE SODIUM (AvPAK), PANTOPRAZOLE SODIUM (AvPAK), PANTOPRAZOLE SODIUM (BE Pharmaceuticals), PANTOPRAZOLE SODIUM (Bryant Ranch Prepack), PANTOPRAZOLE SODIUM (Bryant Ranch Prepack), PANTOPRAZOLE SODIUM (Bryant Ranch Prepack), PANTOPRAZOLE SODIUM (Bryant Ranch Prepack), PANTOPRAZOLE SODIUM (Bryant Ranch Prepack), PANTOPRAZOLE SODIUM (Bryant Ranch Prepack), PANTOPRAZOLE SODIUM (Bryant Ranch Prepack), PANTOPRAZOLE SODIUM (Burel Pharmaceuticals), PANTOPRAZOLE SODIUM (Camber Pharmaceuticals), PANTOPRAZOLE SODIUM (Camber Pharmaceuticals), PANTOPRAZOLE SODIUM (Camber Pharmaceuticals), PANTOPRAZOLE SODIUM (Cardinal Health 107), PANTOPRAZOLE SODIUM (Cardinal Health 107), PANTOPRAZOLE SODIUM (Cardinal Health 107), PANTOPRAZOLE SODIUM (Cardinal Health 107), PANTOPRAZOLE SODIUM (Cardinal Health 107), PANTOPRAZOLE SODIUM (Chartwell RX), PANTOPRAZOLE SODIUM (DIRECT RX), PANTOPRAZOLE SODIUM (Devatis), PANTOPRAZOLE SODIUM (Edenbridge Pharmaceuticals LLC.), PANTOPRAZOLE SODIUM (Eugia US LLC), PANTOPRAZOLE SODIUM (Fresenius Kabi USA), PANTOPRAZOLE SODIUM (Gland Pharma Limited), PANTOPRAZOLE SODIUM (Golden State Medical Supply), PANTOPRAZOLE SODIUM (Granules Pharmaceuticals), PANTOPRAZOLE SODIUM (Lannett Company), PANTOPRAZOLE SODIUM (Major Pharmaceuticals), PANTOPRAZOLE SODIUM (Major Pharmaceuticals), PANTOPRAZOLE SODIUM (Meitheal Pharmaceuticals), PANTOPRAZOLE SODIUM (Meitheal Pharmaceuticals), PANTOPRAZOLE SODIUM (Mylan Institutional), PANTOPRAZOLE SODIUM (Mylan Pharmaceuticals), PANTOPRAZOLE SODIUM (NCS HealthCare of KY), PANTOPRAZOLE SODIUM (NCS HealthCare of KY), PANTOPRAZOLE SODIUM (NorthStar Rx LLC), PANTOPRAZOLE SODIUM (NorthStar RxLLC), PANTOPRAZOLE SODIUM (Northstar Rx LLC), PANTOPRAZOLE SODIUM (Northwind Health Company), PANTOPRAZOLE SODIUM (Northwind Health Company), PANTOPRAZOLE SODIUM (Northwind Health Company), PANTOPRAZOLE SODIUM (Novadoz Pharmaceuticals LLC), PANTOPRAZOLE SODIUM (NuCare Pharmaceuticals), PANTOPRAZOLE SODIUM (NuCare Pharmaceuticals), PANTOPRAZOLE SODIUM (NuCare Pharmaceuticals), PANTOPRAZOLE SODIUM (NuCare Pharmaceuticals), PANTOPRAZOLE SODIUM (NuCare Pharmaceuticals), PANTOPRAZOLE SODIUM (NuCare Pharmaceuticals), PANTOPRAZOLE SODIUM (NuCare Pharmaceuticals), PANTOPRAZOLE SODIUM (PD-Rx Pharmaceuticals), PANTOPRAZOLE SODIUM (PD-Rx Pharmaceuticals), PANTOPRAZOLE SODIUM (PHOENIX RX LLC), PANTOPRAZOLE SODIUM (Piramal Critical Care), PANTOPRAZOLE SODIUM (Preferred Pharmaceuticals), PANTOPRAZOLE SODIUM (Proficient Rx LP), PANTOPRAZOLE SODIUM (Proficient Rx LP), PANTOPRAZOLE SODIUM (Proficient Rx LP), PANTOPRAZOLE SODIUM (Proficient Rx LP), PANTOPRAZOLE SODIUM (Proficient Rx LP), PANTOPRAZOLE SODIUM (QPharma), PANTOPRAZOLE SODIUM (QPharma), PANTOPRAZOLE SODIUM (QPharma), PANTOPRAZOLE SODIUM (Quality Care Products), PANTOPRAZOLE SODIUM (Quallent Pharmaceuticals Health LLC), PANTOPRAZOLE SODIUM (REMEDYREPACK), PANTOPRAZOLE SODIUM (REMEDYREPACK), PANTOPRAZOLE SODIUM (REMEDYREPACK), PANTOPRAZOLE SODIUM (Rising Pharma Holdings), PANTOPRAZOLE SODIUM (Safecor Health LLC), PANTOPRAZOLE SODIUM (Sagent Pharmaceuticals), PANTOPRAZOLE SODIUM (Sandoz), PANTOPRAZOLE SODIUM (St Mary's Medical Park Pharmacy), PANTOPRAZOLE SODIUM (Sun Pharmaceutical Industries Limited), PANTOPRAZOLE SODIUM (Sun Pharmaceutical Industries), PANTOPRAZOLE SODIUM (Sun Pharmaceutical Industries), PANTOPRAZOLE SODIUM (Torrent Pharmaceuticals Limited), PANTOPRAZOLE SODIUM (XLCare Pharmaceuticals), PANTOPRAZOLE SODIUM DELAYED-RELEASE (Cipla USA), PANTOPRAZOLE SODIUM DELAYED-RELEASE (Lifestar Pharma LLC), PANTOPRAZOLE SODIUM DR (Direct_Rx) |
|---|
| 組成式 |
(C16H14F2N3O4S. Na)2. 3H2O
|
|---|
| 質量 |
864.1459
|
|---|
| 分子量 |
864.75
|
|---|
| 構造式 |
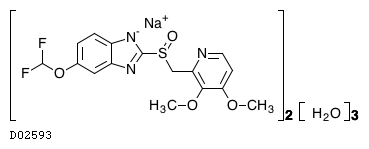
|
|---|
| クラス |
|
|---|
| コード |
|
|---|
| 効能 |
消化性潰瘍薬, プロトンポンプ阻害薬 |
|---|
| コメント |
ベンゾイミダゾール誘導体
|
|---|
| ターゲット |
|
|---|
| パスウェイ |
|
|---|
| 代謝 |
Enzyme: CYP2C19 [HSA: 1557]; CYP3A4 [HSA: 1576]
|
|---|
| 相互作用 |
|
|---|
| 構造マップ |
|
|---|
| 階層分類 |
医療用医薬品のATC分類 [BR:jp08303]
A 消化管と代謝作用
A02 胃酸関連疾患用薬
A02B 消化性潰瘍と胃食道逆流用薬
A02BC プロトンポンプ阻害薬
A02BC02 パントプラゾール
D02593 パントプラゾールナトリウム水和物 (JAN) <US>
医療用医薬品のUSP分類 [BR:jp08302]
胃腸薬
プロトンポンプ阻害薬
パントプラゾール
D02593 パントプラゾールナトリウム水和物 (JAN)
医薬品グループ [BR:jp08330]
消化器系用薬
DG01975 消化性潰瘍薬
DG01646 プロトンポンプ阻害薬 (PPI)
DG00021 パントプラゾール
D02593 パントプラゾールナトリウム水和物
代謝酵素基質薬
DG01639 CYP2C19基質薬
DG00021 パントプラゾール
D02593 パントプラゾールナトリウム水和物
DG01633 CYP3A/CYP3A4基質薬
DG02913 CYP3A4基質薬
DG00021 パントプラゾール
D02593 パントプラゾールナトリウム水和物
ターゲットに基づく医薬品分類 [BR:jp08310]
酵素
ヒドロラーゼ (EC3)
ATPアーゼ
ATP4
D02593 パントプラゾールナトリウム水和物 (JAN) <US>
日本・米国・欧州の新薬 [jp08328.html]
日米欧での承認状況
D02593
薬物代謝酵素とトランスポーター [jp08309.html]
薬物代謝酵素
D02593
ゲノムバイオマーカー [jp08341.html]
医薬品応答に影響を与える多型と変異
D02593
医薬品グループ [BR:jp08330]
消化器系用薬
DG01975 消化性潰瘍薬
DG01646 プロトンポンプ阻害薬 (PPI)
DG00021 パントプラゾール
代謝酵素基質薬
DG01639 CYP2C19基質薬
DG00021 パントプラゾール
DG01633 CYP3A/CYP3A4基質薬
DG02913 CYP3A4基質薬
DG00021 パントプラゾール
|
|---|
| リンク |
|
|---|
| LinkDB |
|
|---|
|
» English version » Back
|